Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 , ThO
, ThO , UO
, UO , PuO
, PuO , and (U, Pu)O
, and (U, Pu)O
Kato, Masato; Watanabe, Masashi; Hirooka, Shun; Vauchy, R.
Frontiers in Nuclear Engineering (Internet), 1, p.1081473_1 - 1081473_10, 2023/01

Watanabe, Masashi; Seki, Takayuki*
Materials Science & Engineering B, 272, p.115369_1 - 115369_6, 2021/10
Times Cited Count:1 Percentile:7.92(Materials Science, Multidisciplinary)The effect of oxygen non-stoichiometry on the initial sintering behavior of CeO was investigated. It was found that the initial sintering of the stoichiometric and hypo-stoichiometric composition was controlled by the grain boundary diffusion. The activation energies of cation diffusion were derived from initial sintering data. Moreover, it is suggested that the cation diffusion was caused by a vacancy mechanism.
was investigated. It was found that the initial sintering of the stoichiometric and hypo-stoichiometric composition was controlled by the grain boundary diffusion. The activation energies of cation diffusion were derived from initial sintering data. Moreover, it is suggested that the cation diffusion was caused by a vacancy mechanism.
Mihara, Morihiro; Harasawa, Shuichi*; Torii, Kazuyuki*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 26(1), p.15 - 23, 2019/06
Hardened cement pastes (HCPs) of water/cement ratio (W/C) 50% and 30% using fly ash (FA), blast furnace slag (BFS) and silica fume (SF) for 28 days were prepared. Apparent diffusion coefficients (D ) of Cs and I in HCPs were obtained using electron probe microanalysis. For Cs, BFS and SF contributed to a reduction of D
) of Cs and I in HCPs were obtained using electron probe microanalysis. For Cs, BFS and SF contributed to a reduction of D for W/C=50% and 30%, respectively. For I, BFS reduced D
for W/C=50% and 30%, respectively. For I, BFS reduced D for W/C=50%, however a significant reduction in D
for W/C=50%, however a significant reduction in D for W/C=30% was not observed. Using SF enhanced sorption of Cs on HCP, and using BFS slightly improved sorption of Cs on HCP. It was also confirmed that the pore structures of HCP using SF and BFS were connected by fine pores. It was therefore considered that using SF and BFS contributes to the reduction of D
for W/C=30% was not observed. Using SF enhanced sorption of Cs on HCP, and using BFS slightly improved sorption of Cs on HCP. It was also confirmed that the pore structures of HCP using SF and BFS were connected by fine pores. It was therefore considered that using SF and BFS contributes to the reduction of D in HCP.
in HCP.
 O in Fe
O in Fe O
O film on Fe formed in an NaOH solution containing oxidants
film on Fe formed in an NaOH solution containing oxidantsHaruna, Takumi*; Miyataki, Yuki*; Hirohata, Yohei*; Shibata, Toshio*; Taniguchi, Naoki; Tachikawa, Hirokazu*
Zairyo To Kankyo, 67(9), p.375 - 380, 2018/09
This research aimed to confirm the formation of Fe O
O film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D
film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D O in the Fe
O in the Fe O
O film at room temperature. The results were obtained as follows. It was confirmed that Fe
film at room temperature. The results were obtained as follows. It was confirmed that Fe O
O film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D
film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D
O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D
O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D O constant. The constant amount of D
O constant. The constant amount of D O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D
O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D
O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D O were obtained to be 5.1
O were obtained to be 5.1 10
10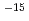 cm
cm s
s and 9.9
and 9.9 10
10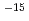 cm
cm s
s for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe
for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe O
O film was in the region from 5.1
film was in the region from 5.1 10
10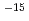 cm
cm s
s to 9.9
to 9.9 10
10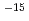 cm
cm s
s .
.
Hamamoto, Takafumi*; Matsubara, Ryuta*; Shibutani, Sanae*; Suyama, Tadahiro*; Tachi, Yukio
JAEA-Data/Code 2017-014, 31 Pages, 2018/03
NUMO and JAEA have developed the methodology of post-closure safety assessment for the geological disposal. For this purpose, NUMO and JAEA have conducted a collaborative research project for developing the safety assessment methodology based on international state of the art knowledge. The present report focuses on investigation of sorption and diffusion data reported and their QA evaluation for updating sorption and diffusion database (SDB and DDB) as the collaborative research project between NUMO and JAEA. This report includes sorption and diffusion data for mainly sedimentary rocks and cement materials. As a result, 1,746 sorption data from 19 references and 593 diffusion data from 25 references were extracted and prepared in the datasheet of SDB and DDB.
Ogino, Masataka*; Owaki, Eiji*; Shirase, Mitsuyasu*; Nakayama, Masashi
Konkurito Kogaku Nenji Rombunshu (DVD-ROM), 39(1), p.703 - 708, 2017/07
no abstracts in English

Watanabe, Masashi; Sunaoshi, Takeo*; Kato, Masato
Defect and Diffusion Forum, 375, p.84 - 90, 2017/05
The oxygen chemical diffusion coefficient in (U, Pu)O was determined by thermo-gravimetry as functions of the Pu content, oxygen-to-metal ratio and temperature. The surface reaction was considered in the diffusion coefficient determination. The activation energy for the chemical diffusion coefficient was 60 kJ/mol and 65 kJ/mol, respectively, in (U
was determined by thermo-gravimetry as functions of the Pu content, oxygen-to-metal ratio and temperature. The surface reaction was considered in the diffusion coefficient determination. The activation energy for the chemical diffusion coefficient was 60 kJ/mol and 65 kJ/mol, respectively, in (U Pu
Pu )O
)O and (U
and (U Pu
Pu )O
)O .
.
 -Iron Using First-Principles-Based Kinetic Monte Carlo
-Iron Using First-Principles-Based Kinetic Monte CarloEbihara, Kenichi; Suzudo, Tomoaki; Yamaguchi, Masatake
Materials Transactions, 58(1), p.26 - 32, 2017/01
Times Cited Count:8 Percentile:38.56(Materials Science, Multidisciplinary)In order to evaluate grain boundary (GB) phosphorous (P) segregation in nuclear reactor pressure vessel steels under irradiation, the rate-theory model based on first-principles calculations is developed. In this study, we evaluated the diffusion coefficient of the mixed interstitial dumbbell of a P atom and an iron(Fe) atom using a kinetic Monte Carlo (kMC) simulation based on first-principles calculations. The evaluated diffusion coefficient was almost the same with the diffusion coefficient of P atoms which migrate via octahedral interstitial sites, and was much faster than that for P transport by vacancies. Furthermore, from the simulation of the irradiation induced GB P segregation using the model which was modified to include P atoms of octahedral interstitial sites, it was found that the boundary condition at GB is not valid for P atoms of octahedral interstitial sites
Sato, Tomonori; Yamamoto, Masahiro; Tsukada, Takashi; Kato, Chiaki
Zairyo To Kankyo, 64(3), p.91 - 97, 2015/03
In the boiling water reactors (BWRs), reactor cooling water is maintained in high purity condition by controlling of a deionizing and deaerating apparatus, however H O
O contains by water radiolysis. In order to determine the corrosive condition in high-temperature pure water containing H
contains by water radiolysis. In order to determine the corrosive condition in high-temperature pure water containing H O
O , the electrochemical impedance spectroscopy was performed in this study. To simulate BWR condition precisely, the measurements were performed without any electrolyte. The obtained impedance responses were changed with the H
, the electrochemical impedance spectroscopy was performed in this study. To simulate BWR condition precisely, the measurements were performed without any electrolyte. The obtained impedance responses were changed with the H O
O concentration. The solution resistance and polarization resistance were determined by the equivalent circuit analyses. The conductivity was determined by the obtained solution resistance and the calculation of the current flow between the working electrode and the counter electrode by the 3-demensional finite element method. The value, 4.4
concentration. The solution resistance and polarization resistance were determined by the equivalent circuit analyses. The conductivity was determined by the obtained solution resistance and the calculation of the current flow between the working electrode and the counter electrode by the 3-demensional finite element method. The value, 4.4 10
10 S/cm, was obtained as the conductivity of the pure water at 288
S/cm, was obtained as the conductivity of the pure water at 288 C. The reciprocal of the obtained polarization resistance increased in proportion with H
C. The reciprocal of the obtained polarization resistance increased in proportion with H O
O concentration. This indicates that the corrosion current density was determined by the diffusion limiting current density of H
concentration. This indicates that the corrosion current density was determined by the diffusion limiting current density of H O
O . The diffusion coefficient of H
. The diffusion coefficient of H O
O at 288
at 288 C was determined using the relationship between the reciprocal of the polarization resistance and H
C was determined using the relationship between the reciprocal of the polarization resistance and H O
O concentration. The obtained diffusion coefficient was 1.5
concentration. The obtained diffusion coefficient was 1.5 10
10 cm
cm /s. This is about twice larger than that of the reported value of O
/s. This is about twice larger than that of the reported value of O .
.
 -ray irradiation on corrosion of low alloy steel in neutral water
-ray irradiation on corrosion of low alloy steel in neutral waterYamamoto, Masahiro; Nakano, Junichi; Komatsu, Atsushi; Sato, Tomonori; Tsukada, Takashi
Proceedings of 19th International Corrosion Congress (19th ICC) (CD-ROM), 6 Pages, 2014/11
Corrosion protection of RPV and PCV is an important issue for the long term maintenance until the end of the decommissioning procedures. One of the uncertain factors for the issue is an effect of radioactivity on corrosion of LAS and CS. Corrosion tests using LAS and CS were conducted in  -rays irradiated condition. Oxygen and hydrogen peroxide concentrations in the water were measured after the tests. Corrosion test results indicated that the amounts of corrosion increased by
-rays irradiated condition. Oxygen and hydrogen peroxide concentrations in the water were measured after the tests. Corrosion test results indicated that the amounts of corrosion increased by  -rays irradiation both air and nitrogen atmosphere. And also corrosion amounts increased with
-rays irradiation both air and nitrogen atmosphere. And also corrosion amounts increased with  -ray dose rates. Electrochemical analyses indicated that cathodic reaction of Hydrogen peroxide was controlled by diffusion process. The measured diffusion constant of H
-ray dose rates. Electrochemical analyses indicated that cathodic reaction of Hydrogen peroxide was controlled by diffusion process. The measured diffusion constant of H O
O was about 0.75 times to that of oxygen. From these results, it is estimated that corrosion of LAS and CS in
was about 0.75 times to that of oxygen. From these results, it is estimated that corrosion of LAS and CS in  -ray irradiated condition was evaluated by the cathodic reduction reaction of oxidant.
-ray irradiated condition was evaluated by the cathodic reduction reaction of oxidant.
Inagaki, Shigeru*; Takenaga, Hidenobu; Ida, Katsumi*; Isayama, Akihiko; Tamura, Naoki*; Takizuka, Tomonori; Shimozuma, Takashi*; Kamada, Yutaka; Kubo, Shin*; Miura, Yukitoshi; et al.
Nuclear Fusion, 46(1), p.133 - 141, 2006/01
Times Cited Count:54 Percentile:85.24(Physics, Fluids & Plasmas)no abstracts in English
Yamaguchi, Tetsuji; Sakamoto, Yoshifumi; Iida, Yoshihisa; Negishi, Kumi; Taki, Hiroshi; Akai, Masanobu; Jinno, Fumika; Kimura, Yuichiro; Ueda, Masato; Tanaka, Tadao; et al.
Proceedings of International Conference on Nuclear Energy System for Future Generation and Global Sustainability (GLOBAL 2005) (CD-ROM), 6 Pages, 2005/10
no abstracts in English
Yamaguchi, Tetsuji; Minase, Naofumi; Iida, Yoshihisa; Tanaka, Tadao; Nakayama, Shinichi
JAERI-Conf 2005-007, p.150 - 155, 2005/08
no abstracts in English
Hidaka, Akihide*; Kudo, Tamotsu; Kida, Mitsuko; Fuketa, Toyoshi
JAERI-Research 2005-001, 67 Pages, 2005/02
In the VEGA program to investigate radionuclides release from irradiated fuel during severe accidents, the analyses are being performed with VICTORIA2.0 code for comprehensive understanding of radionuclides release and transport phenomena. The VEGA-1 and -3 tests were analyzed in the present study. The correlation for Cs diffusion coefficient in fuel grain obtained from VEGA-1 was applied to the release analysis of VEGA-3. The calculated release of Cs agreed well with the measurement. The correlation was applied to subsequent Cs transport and deposition analyses. The calculation underpredicted the total mass of Cs deposited onto the test apparatuses because nucleation of aerosol and its growth were underestimated due to the consideration of aerosol nucleation originated only from released FP in VICTORIA2.0. A sensitivity analysis with aerosol seeds for heterogeneous nucleation showed a reasonable agreement with the measured Cs distribution. It turned out that additional aerosol seeds besides the released FP be considered when the VICTORIA2.0 code is applied to the VEGA test analyses.
Takenaga, Hidenobu; Oyama, Naoyuki; Isayama, Akihiko; Inagaki, Shigeru*; Takizuka, Tomonori; Fujita, Takaaki; Miura, Yukitoshi
Europhysics Conference Abstracts (CD-ROM), 29C, 4 Pages, 2005/00
no abstracts in English
Inagaki, Shigeru*; Takenaga, Hidenobu; Ida, Katsumi*; Isayama, Akihiko; Tamura, Naoki*; Takizuka, Tomonori; Shimozuma, Takashi*; Kamada, Yutaka; Kubo, Shin*; Miura, Yukitoshi; et al.
Proceedings of 20th IAEA Fusion Energy Conference (FEC 2004) (CD-ROM), 8 Pages, 2004/11
no abstracts in English
Sakamoto, Yoshiteru; Suzuki, Takahiro; Ide, Shunsuke; Koide, Yoshihiko; Takenaga, Hidenobu; Kamada, Yutaka; Fujita, Takaaki; Fukuda, Takeshi; Takizuka, Tomonori; Shirai, Hiroshi; et al.
Nuclear Fusion, 44(8), p.876 - 882, 2004/08
Times Cited Count:33 Percentile:71.1(Physics, Fluids & Plasmas)Response of the ion thermal diffusivity to the radial electric field Er shear has been investigated in JT-60U and the following results were found. (1) In the case of positive magnetic shear (PS) plasma, the ion thermal diffusivity in the core region shows L mode state, weak internal transport barrier (ITB), and strong ITB depending upon the heating power. In the case of reversed magnetic shear (RS) plasma, however, no power degradation of the ion thermal diffusivity is observed. (2) In the case of weak ITB, the ion thermal diffusivity decreases gradually with increasing the Er shear for both PS and RS plasmas. There exists a threshold of an effective Er shear to change its state from weak to strong ITBs. (3) The threshold of the effective Er shear in the case of RS plasma is small compared with that in the case of PS plasma.
Fujita, Takaaki; Fukuda, Takeshi*; Sakamoto, Yoshiteru; Ide, Shunsuke; Suzuki, Takahiro; Takenaga, Hidenobu; Ida, Katsumi*; Idei, Hiroshi*; Shimozuma, Takashi*; Fujisawa, Akihide*; et al.
Plasma Physics and Controlled Fusion, 46(5A), p.A35 - A43, 2004/05
Times Cited Count:26 Percentile:63.74(Physics, Fluids & Plasmas)no abstracts in English
Takenaga, Hidenobu; Oyama, Naoyuki; Fujita, Takaaki; Yamada, Hiroshi*; Nishimura, Kiyohiko*; Tanaka, Kenji*; Sakamoto, Ryuichi*
Annual Report of National Institute for Fusion Science; April 2003 - March 2004, P. 12, 2003/10
no abstracts in English
Yamaguchi, Tetsuji; Nakayama, Shinichi
JAERI-Conf 2003-018, p.115 - 116, 2003/10
Effective diffusivity was determined for Np(IV) in bentonite material in presence of carbonate and for Pu(IV) in presence of fulvic acid. Adsorption of actinides onto negatively charged mineral surfaces were investigated under conditions that actinides were predominantly present as anionic complex species. The results of this study will reveal behavior of actinides under various geological environments.